Overview
ValidLabels is a regulatory SaaS platform built by Awakast in close partnership with AFL Laboratories — the regulatory experts behind ELiX. AFL brought deep domain knowledge of CLP compliance, market surveillance, and chemical safety workflows; Awakast translated that expertise into production-grade software architecture. The result is a platform that handles the full lifecycle of multilingual CLP labels: translation, AI validation, version control, and direct packing-line print integration.
The challenge
CLP compliance requires precise translation of hazard statements, precautionary phrases, and pictogram assignments into every language of the target market — and that set is not static. 29 regulatory updates since 2008. A May 2026 CLP revision deadline. Fines up to €75,000 per violation. Traditional regulatory consultants charged around €40 per label update, turnaround took days, and with portfolios of 1,200+ SKUs across 10+ EU markets, the math was brutal: managing labels manually meant either enormous cost, enormous risk, or both.

Our approach
Awakast designed and shipped ValidLabels end-to-end: a multi-tenant SaaS platform with domain-fine-tuned AI translation modules, sentence-level CLP phrase validation, version-controlled label repository, Zebra ZPL print integration, and team-based audit workflows. LLM support handles free-text safety descriptions with domain-constrained prompting, while custom AI modules enforce regulatory phrase correctness at a sentence level. OAuth 2.0 secures multi-user access with role-based permissions. All infrastructure is self-hosted on Kubernetes with EU data residency — label data never leaves client-controlled environments.
What we delivered
- •Multi-tenant SaaS platform with team permissions and OAuth 2.0 SSO
- •Domain-fine-tuned AI translation modules for sentence-level CLP phrase mapping
- •LLM-powered free-text safety description translation with regulatory guardrails
- •Zebra ZPL print-on-demand integration for sub-7-second scan-to-print on packing lines
- •Version history and full audit log for every label modification and approval
- •Centralized SDS and label repository with role-based access control
- •Automated compliance flagging when CLP regulation updates affect saved labels
Architecture and implementation
- •Custom AI modules trained on CLP regulatory corpus — not general-purpose translation; phrase accuracy validated against Annex III/VI terminology
- •LLM layer with domain-constrained prompting for free-text product descriptions, followed by a validation step before label commit
- •Self-hosted Kubernetes cluster ensuring full EU data residency — no label data reaches third-party infrastructure
- •ZPL output engine compatible with Zebra ZD421, end-to-end scan-to-print tested at under 7 seconds on live packing lines
- •PostgreSQL with immutable label version history — every sentence change is timestamped and attributed for market surveillance and regulatory audit response
- •OAuth 2.0 with RBAC: editor, validator, and admin permission scopes with full activity logging
Delivery timeline
Phase 1
Regulatory domain research
Deep mapping of CLP Regulation (EC) No 1272/2008, hazard phrase taxonomy, multilingual phrase annex, and market-specific labelling edge cases.
Phase 2
Core platform & data model
Multi-tenant architecture, label repository, PostgreSQL versioned schema, OAuth 2.0 integration, and team permission model.
Phase 3
AI translation engine
Custom AI modules for CLP phrase matching, LLM integration for free-text with regulatory constraints, and full validation pipeline build-out.
Phase 4
Print integration & production pilot
Zebra ZPL output engine, packing-line timing validation, ELiX pilot with 1,200+ SKU portfolio, and production hardening.
Screens and artifacts
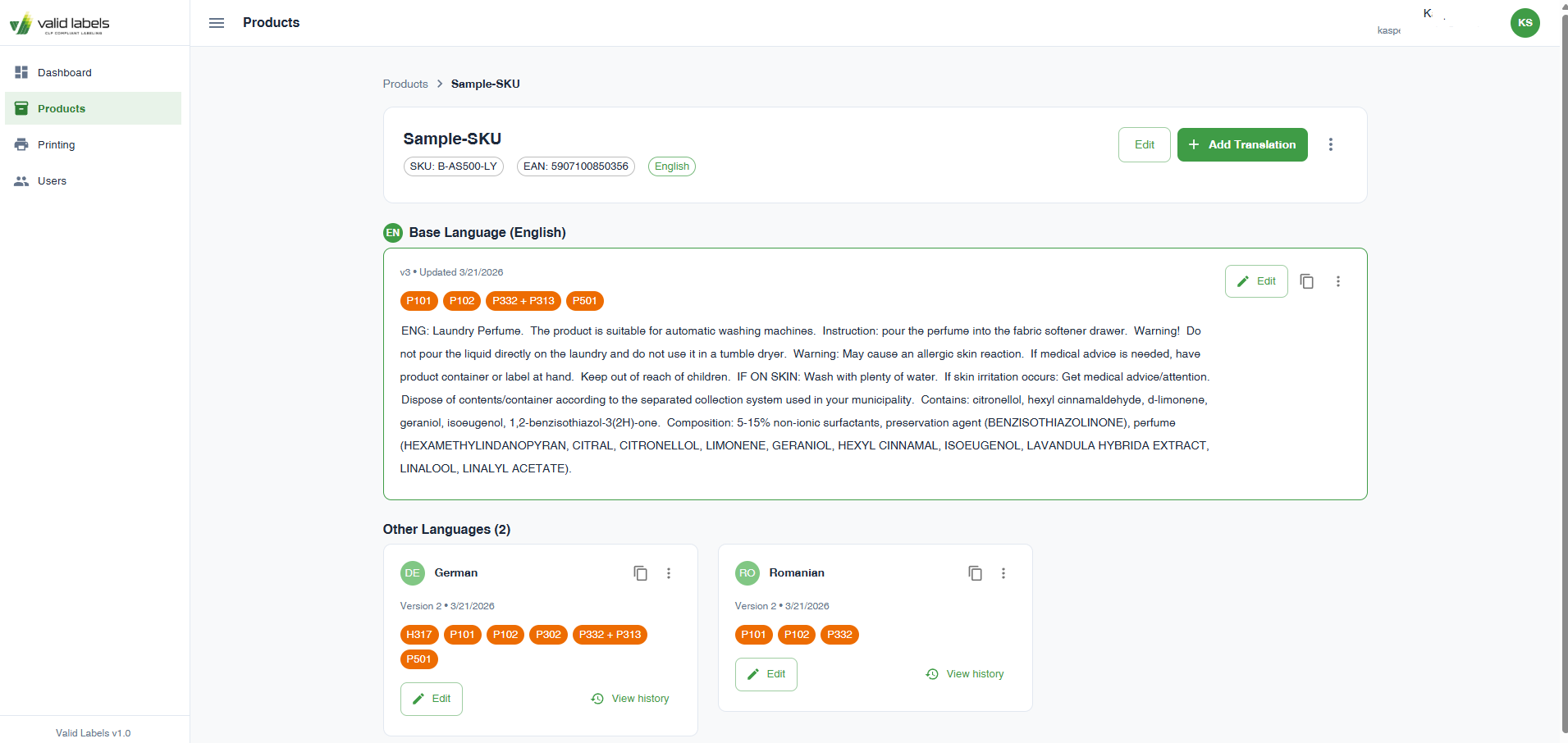
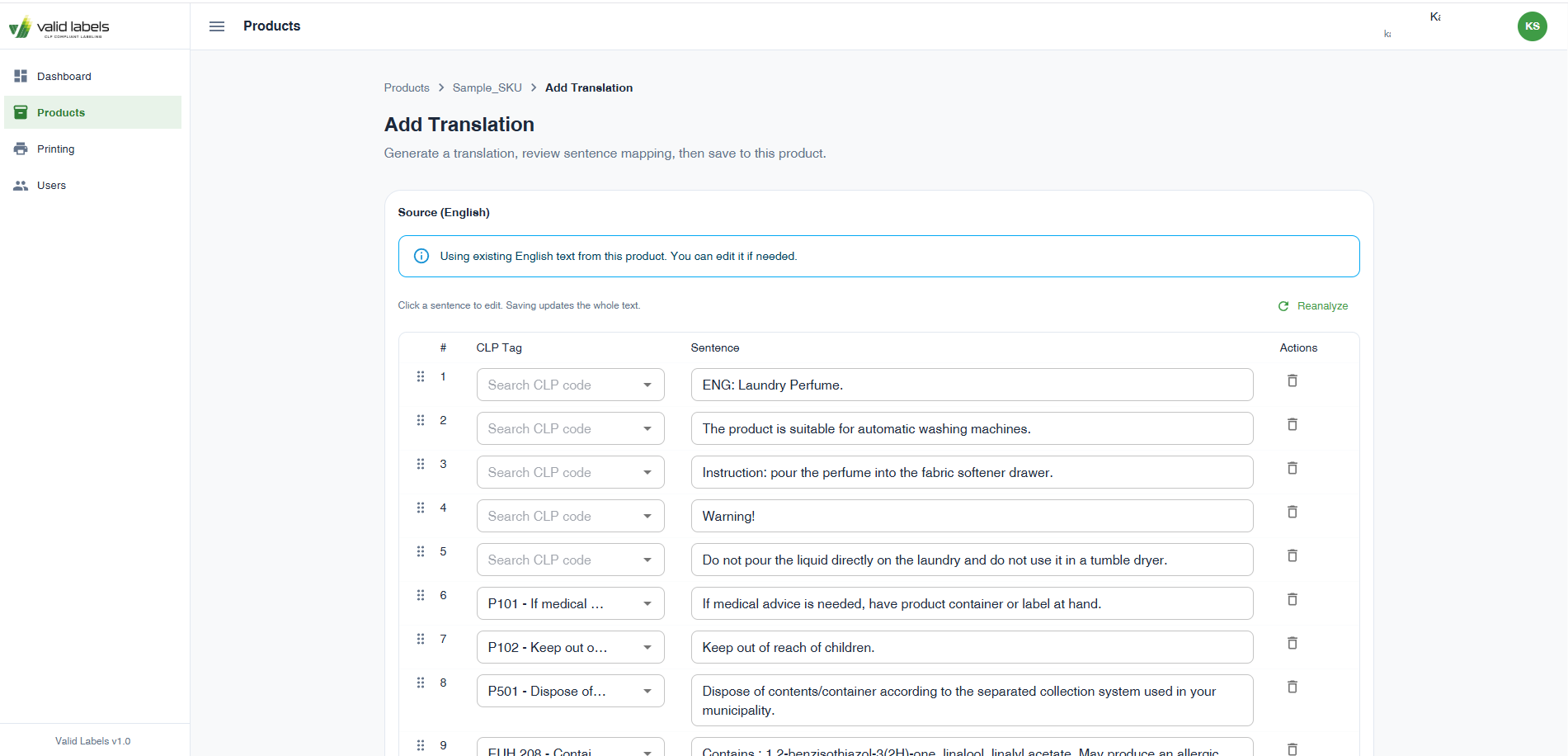
Cost per label translation
Technology
Compliance and controls
- All label data processed on self-hosted EU infrastructure — no third-party data transfer
- Full version history enables market surveillance response and regulatory audit within minutes
- CLP phrase validation at commit prevents non-compliant label states from reaching print or export
Key outcomes
- ✓Regulatory-accurate CLP labels generated in minutes instead of days — without regulatory consultants
- ✓Direct packing-line ZPL integration eliminated manual label handling on the production floor
- ✓Audit trail and version history built for market surveillance, regulatory inspections, and traceability
"We didn't expect the AI to get CLP phrase accuracy right that fast. No revision rounds, no terminology corrections — it just worked."
AFL Laboratories
